 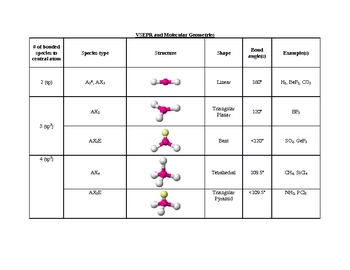
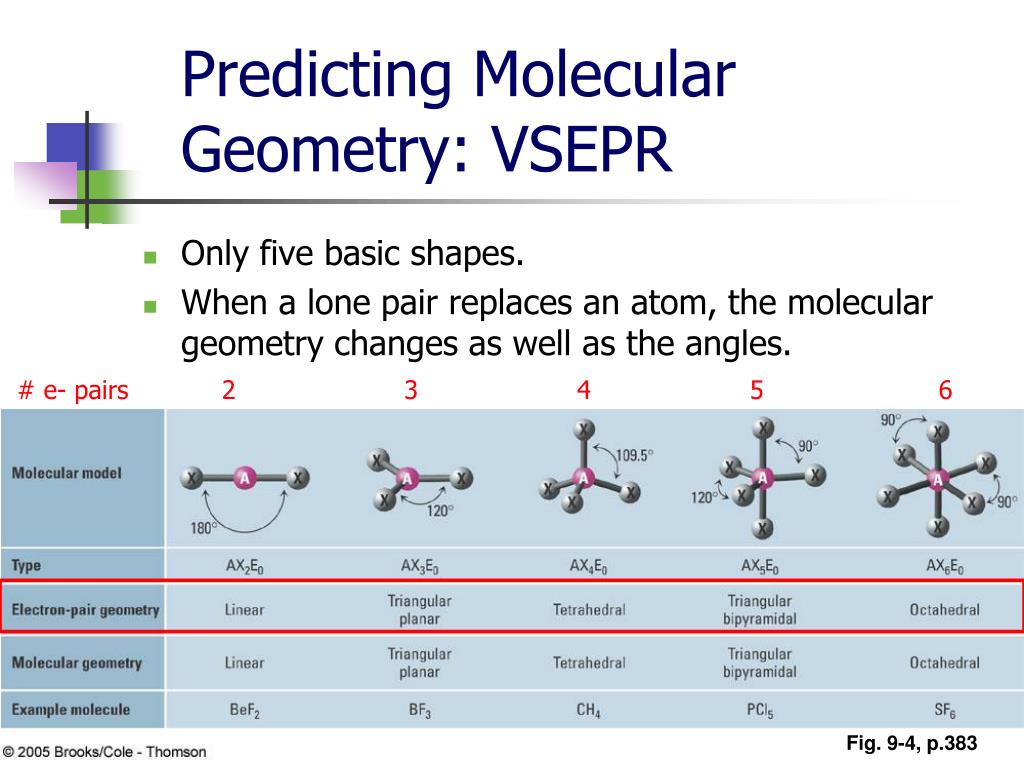
The four equivalent bonds point in four geometrically equivalent directions in three dimensions, corresponding to the four corners of a tetrahedron centered on the carbon atom. An example of a tetrahedral molecule is methane (CH 4). In accordance with the VSEPR theory, the bond angles between the electron bonds are 109.5 o. " This shape is found when there are four bonds all on one central atom, with no lone electron pairs. Tetra- signifies four, and -hedral relates to a face of a solid "tetrahedral" literally means "having four faces. The repulsion between these will be at a minimum when the angle between any two is 120 o. An AX 3 molecule such as BF 3 has three regions of electron density extending out from the central atom. Molecules with the trigonal planar shape are triangular and in one plane, or flat surface. Lewis dot structure of carbon dioxide: Although the central atom (carbon) has four bonds, only two are sigma bonds it is therefore is represented as AX 2E 0 in the table. VSEPR uses the steric number and distribution of X's and E's to predict molecular geometric shapes. The sum of X and E, known as the steric number, is also associated with the total number of hybridized orbitals used by valence bond theory. A in AXE represents the central atom and always has an implied subscript one X represents the number of sigma bonds between the central and outside atoms (multiple covalent bonds-double, triple, etc.- count as one X) and E represents the number of lone electron pairs surrounding the central atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
